- Everyday Alpha
- Posts
- The Kidney Drug That Just Turned Proof into Momentum
The Kidney Drug That Just Turned Proof into Momentum
A blockbuster earnings surprise, a fast-scaling rare disease drug, and a second growth leg just opening. This is where biotech stories stop being speculative and start getting revalued.
It doesn't take much to change the narrative, proof that something is working at scale. That’s exactly what just showed up here, and the market is starting to react.

Tax Strategy (Sponsored)
Many investors overlook deductions that could help minimize capital gains tax, such as:
Eligible investment expenses
Cost basis adjustments
Selling costs tied to property
Each comes with IRS rules and reporting requirements.
That’s why consulting a fiduciary financial advisor is often recommended.

Travere Therapeutics, Inc.

May 6 – Pre‑market
Ticker: TVTX | Sector: Biotechnology / Healthcare | Market Cap: $4.1B

30‑Second Take
Travere just flipped from "interesting pipeline story" to "commercial execution story that's working." FILSPARI is now doing real numbers, hitting over $100M in quarterly sales with strong growth and adoption.
At the same time, the FSGS approval opens a second leg of expansion on top of an already underpenetrated market.
What makes this setup compelling is the combination. You’ve got a drug already scaling in IgAN, a fresh approval unlocking a new patient pool, and a pipeline asset behind it that could extend the story again. This is no longer about whether the science works. It’s about how big this can get if execution holds.

Trade Setup
Timeframe: Long term
Edge type: Commercial execution meets expanding indication
The setup is straightforward but powerful. Travere is moving from a single-indication growth story into a multi-indication rollout, with FILSPARI already proving it can scale and now expanding into FSGS with a ready-built commercial engine behind it.
If the company continues to execute on uptake while layering in this second growth driver, the market has room to reprice this from a “one-product risk” biotech into a durable rare disease franchise.

Gold Protection Play (Sponsored)
Tensions in Iran are escalating, not cooling down. Airstrikes continue, oil routes are under pressure, and analysts warn the economic fallout could deepen.
Gas prices are already climbing. Inflation could spike again. And portfolios heavy in stocks and bonds may be exposed if this situation worsens.
That's why many Americans are moving a portion of their retirement savings into physical gold — an asset that historically holds value during periods of conflict and uncertainty.
Red State Gold Group's FREE Gold IRA Guide shows how eligible IRA or 401(k) funds can be rolled into physical gold and silver.
Or call (888) 711-2433 for more information.

How much did Americans spend on their pets in 2023 — more than the GDP of many countries? |

Snapshot Table
Metric | Value | Current Stance |
|---|---|---|
Price | $44.80 | Below average |
52‑week range | $13.88 - $45.43 | Below average |
Short interest | 12.13% | Below average |
Next catalyst | FSGS uptake data |

Chart
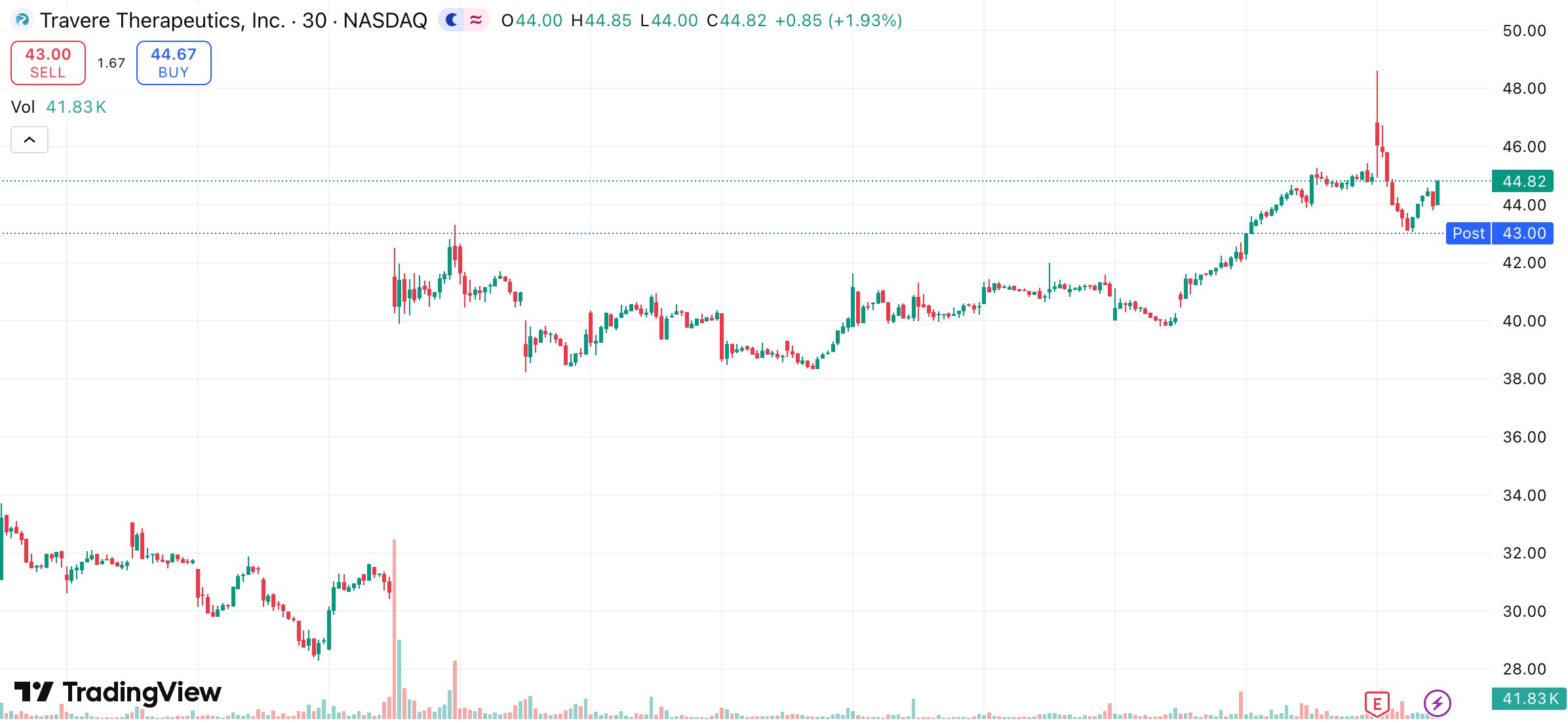
1-month trading summary: TVTX spent the early part of the month drifting lower before everything changed on earnings. A 207% EPS surprise acted as the reset moment, triggering a sharp repricing as the market caught up to how strong the underlying execution actually is.
What stands out is what happened next. The stock didn’t spike and fade. It held those gains, consolidated, and then pushed higher again, now sitting up around 47% on the month. That kind of follow-through points to accumulation, not just a short-term reaction.

Bull Case
From proving it to scaling it: At the center of this story is FILSPARI, Travere’s kidney disease drug designed to reduce protein leakage and slow the decline in kidney function in rare conditions like IgA nephropathy and FSGS. These are serious diseases that often lead to dialysis or transplant, which makes an effective, long-term treatment highly valuable.
What matters now is that this is already working in the real world. FILSPARI is generating meaningful revenue, adoption is building, and the company has clearly shown it can execute commercially.
This is no longer a "wait for approval" biotech story. It's a live, scaling product gaining traction with the doctors who matter.
The model itself is what makes this interesting. Rare disease markets are small but deep, and once a drug becomes embedded with specialists, prescribing tends to build steadily rather than spike and fade. That creates a more durable growth curve than most biotech launches.
This is where the shift happens. When a drug proves it can scale, the conversation moves from potential to durability. Travere is now in that phase, and the market is still catching up to what that can look like over time.
Execution turning into acceleration: The FSGS rollout is the immediate driver. Same drug, same specialists, new patients. If uptake builds as expected, forecasts move higher.
Guidelines and low penetration add fuel. As prescribing becomes more routine, growth exceeds market expectations.
The pipeline adds a second layer. The HARMONY study, a late-stage trial for Travere’s next potential drug in a rare metabolic disease, is progressing and gives the story another leg beyond FILSPARI.
Analyst price targets point to upside: Wall Street is pricing TVTX between $40.00 and $56.00, reflecting confidence in continued execution but still leaving room for upside if the rollout keeps delivering.
Momentum backed by real buying: The breakout post-earnings and tight consolidation that followed point to accumulation, not a one-day reaction. As long as TVTX holds these higher levels, the trend is working in its favor.

Bear Case
Execution has to stay clean: Travere’s success rides or fails on continued commercial delivery. If FILSPARI uptake slows, particularly in the early stages of the FSGS rollout, the growth narrative could lose momentum quickly.
Another risk is concentration. With most of the value tied to a single drug, any setback on safety, reimbursement, or physician adoption will have an outsized impact.
Competition is building in the background: Travere has a first-mover advantage, but it won't have the field to itself. Larger players and newer biotech names are targeting the same kidney disease pathways, including immunosuppressive therapies, complement inhibitors, and next-gen targeted drugs.
That matters because some of these competitors have deeper pipelines, larger balance sheets, and established relationships with nephrologists. If a rival therapy shows stronger outcomes or easier use, it can quickly chip away at share in what is still an evolving treatment landscape.
Biotech sentiment and pricing pressure still matter: Even with strong execution, Travere sits in a sector that can turn quickly. Biotech sentiment is highly sensitive to rates, risk appetite, and capital flows, which can cap upside even as fundamentals improve.
There’s also the ever-present pressure on drug pricing and reimbursement. Rare disease therapies command premium pricing, but that scrutiny is increasing. Any pushback from payers or policy shifts can slow adoption or compress expectations.
Still early, not overcrowded: Despite the recent run, this doesn’t feel like a crowded trade yet. The move has been driven by fundamentals rather than hype, and positioning still looks early in the rerating cycle.
That said, if momentum continues and more investors pile in on the same growth narrative, this can tighten quickly. For now, it looks like accumulation, not excess.

Quick Checklist
✅ Thesis still valid after today’s close
✅ Volume confirms move above key levels
✅ Catalyst date double-checked (May 06, 2026)

Deep‑Dive Links

That’s all for today’s Everyday Alpha. We’ll have a new pick for you every morning before the market opens, so stay tuned!
Best Regards,
—Noah Zelvis
Everyday Alpha

